علم الكيمياء

تاريخ الكيمياء والعلماء المشاهير

التحاضير والتجارب الكيميائية

المخاطر والوقاية في الكيمياء

اخرى

مقالات متنوعة في علم الكيمياء

كيمياء عامة


الكيمياء التحليلية

مواضيع عامة في الكيمياء التحليلية

التحليل النوعي والكمي

التحليل الآلي (الطيفي)

طرق الفصل والتنقية


الكيمياء الحياتية

مواضيع عامة في الكيمياء الحياتية

الكاربوهيدرات

الاحماض الامينية والبروتينات

الانزيمات

الدهون

الاحماض النووية

الفيتامينات والمرافقات الانزيمية

الهرمونات


الكيمياء العضوية

مواضيع عامة في الكيمياء العضوية

الهايدروكاربونات

المركبات الوسطية وميكانيكيات التفاعلات العضوية

التشخيص العضوي

تجارب وتفاعلات في الكيمياء العضوية


الكيمياء الفيزيائية

مواضيع عامة في الكيمياء الفيزيائية

الكيمياء الحرارية

حركية التفاعلات الكيميائية

الكيمياء الكهربائية


الكيمياء اللاعضوية

مواضيع عامة في الكيمياء اللاعضوية

الجدول الدوري وخواص العناصر

نظريات التآصر الكيميائي

كيمياء العناصر الانتقالية ومركباتها المعقدة


مواضيع اخرى في الكيمياء

كيمياء النانو

الكيمياء السريرية

الكيمياء الطبية والدوائية

كيمياء الاغذية والنواتج الطبيعية

الكيمياء الجنائية


الكيمياء الصناعية

البترو كيمياويات

الكيمياء الخضراء

كيمياء البيئة

كيمياء البوليمرات

مواضيع عامة في الكيمياء الصناعية

الكيمياء التناسقية

الكيمياء الاشعاعية والنووية
Anticancer Drugs
المؤلف:
Geoffrey A. Lawrance
المصدر:
Introduction to Coordination Chemistry
الجزء والصفحة:
P252-255
2026-04-01
46
Anticancer Drugs
Cancer, as one of our most significant diseases, is the focus of intensive international study and treatment development. It is interesting to discover that platinum-based chemothera- peutic drugs are amongst the most active and commonly used clinical agents for treating a range of advanced cancers. Diamminedi chloro platinum (II) (called cisplatin in medical use; Figure 9.1) was the first to be used clinically, and remains one of the largest-selling drugs on the market. This is an old coordination compound, first reported by Peyrone in 1845 with its structure defined by Werner. The potential anticancer properties of cisplatin were discovered serendipitously by Rosenberg in 1965 and the drug was introduced clinically in 1971. It is routinely widely used, including for treating ovarian testicular, bladder, head, neck and small-cell lung carcinomas. In particular, cisplatin has made testicular cancer eminently curable, with more than 90% of sufferers now cured (a celebrated case being American cyclist Lance Armstrong a multiple Tour de France winner).
Cisplatin acts by binding to DNA and inhibiting replication in the cancer cell. Substitution reactions of coordinated chloride ligands are the key chemistry in reactions of cisplatin in a human cell. Most cisplatin circulates in the blood unchanged over a short timeframe as
Figure 9.1
The drug cisplatin, its reaction pathways in the cell and an example of coordination to DNA, leading to distortion of the double helix (cisplatin-DNA binding figure reproduced from the Protein Data Bank; DOI:10.2210/pdblaio/pdb).
the high chloride ion concentration in blood suppresses coordinated chloride hydrolysis and hence further reaction. Once across the cytoplasmic membrane of a cell, however, it meets a much reduced chloride concentration (as low as 4 nM) and undergoes a series of hydrolysis reactions, forming species including [PtCl(NH3)2(OH)], where one chloride ligand has been replaced in a substitution reaction by a water ligand. The biological target of the complex is DNA in the cancer cell; however, it is not particularly discriminating in its chemistr, and can participate in coordination chemistry with sulfur-containing groups and other functionalities that lead to its 'capture' and inactivation; it binds so strongly to these that it is no longer available for further chemistry. The interaction sought (Figure 9.1) is with the heterocyclic nitrogen bases that form part of DNA - cytosin, guanine, adenine and thymidine. These types of N-donors are excellent ligands for platinum(II); guanine is believed to be a particularly favourable donor. Binding is covalent - coordinated chloride ion hydrolysis is necessary first, to provide a reasonably labile site (coordinated water) for substitution chemistry that leads to the introduction of the N-donor DNA base into the inner coordination sphere. The cationic aqua complexes formed following initial chloride hydrolysis (particularly [Pt (NH3)2(OH2)2]2+) enhance the activity of the drug as a cytostatic agent due to an ionic attraction to the negatively-charged DNA helix, which has an anionic phosphate backbone. However, it is the structural changes arising from coordination of N-donor DNA bases in place of water ligands that are the key to their activity.
In addition, the cisplatin may bind to other proteins and biomolecules, sometimes together with binding to DNA. In achieving binding to two bio-sites, it is using both cis coordination sites originally occupied by chloride ions, so that hydrolysis of both chloride ion ligands is necessary. Possible binding modes are shown in Figure 9.2. Which of these is most important is under continuing debate but intrastrand processes are favoured. Cisplatin
Figure 9.2 Various modes of coordination of the platinum(II) drug cisplatin to bases in DNA strands, following initial chloride hydrolysis; a guanine N-donor (G) is favoured. Actual models determined from structural studies of intra- (at left) and inter-strand (at right) coordination to DNA oligomers (drawn from the Protein Data Bank; DOI: 10.2210/pdb1ddp/pdb and 10.2210/pdb1ksb/pdb) are also shown.
can form intrastrand crosslinks between adjacent bases, preferring two guanine bases (a cisplatin-guanine-guanine intrastrand DNA adduct is depicted in Figure 9.1). This coordination mode cannot be adopted by inactive transplatinFigure 9.2 Various modes of coordination of the platinum(II) drug cisplatin to bases in DNA strands, following initial chloride hydrolysis; a guanine N-donor (G) is favoured. Actual models determined from structural studies of intra- (at left) and inter-strand (at right) coordination to DNA oligomers (drawn from the Protein Data Bank; DOI: 10.2210/pdb1ddp/pdb and 10.2210/pdb1ksb/pdb) are also shown.
can form intrastrand crosslinks between adjacent bases, preferring two guanine bases (a cisplatin-guanine-guanine intrastrand DNA adduct is depicted in Figure 9.1). This coordination mode cannot be adopted by inactive transplatin, suggesting that it is indeed of importance. This crosslinking has been shown to cause unwinding and duplex bending, perhaps sufficient to attract high-mobility group damage-recognition proteins, which bind and further inhibit replication.
The core problem with cisplatin is that dose-related toxicity occurs, leading to an array of problems for patients, such as joint pain, ringing in the ears and hearing difficulties, and general weakness. The maximum tolerated dose of cisplatin is around 100 mg per day for up to only five consecutive days. While the drug displays high activity, these unfortunate side- effects led to a search for a better analogue (Figure 9.3). Carboplatin [cis-diammine(1,1- cyclobutanedicarboxylato)platinum(II)] was introduced as a second-generation drug in 1990. It shows about a fourfold reduction in side effects to the kidneys and nervous system. Because the drugs must circulate in the bloodstream before finding their target, slower degradation in vivo is believed to be one factor of importance. Both cisplatin and carboplatin suffer from the emergence of acquired resistance with time. Subsequently, oxaliplatin [(1.,2- cyclohexane diamine) (oxalato)platinum(II)] was introduced as a third-generation drug. It is active in all phases of the cell cycle, and binds covalently to DNA guanine and adenine bases dominantly via intrastrand crosslinking. It is thought that DNA mismatch repair enzymes are unable to recognize oxaliplatin-DNA adducts in contrast with some other platinum-DNA adducts, as a result of the bulkier nature of the complex. The complex is inactivated by thiol protein binding, alteration in cellular transport and increased DNA repair enzyme activity. Development of a new-generation drug to address inactivation has led to satraplati the suggesting that it is indeed of importance. This crosslinking has been shown to cause unwinding and duplex bending, perhaps sufficient to attract high-mobility group damage-recognition proteins, which bind and further inhibit replication.
The core problem with cisplatin is that dose-related toxicity occurs, leading to an array of problems for patients, such as joint pain, ringing in the ears and hearing difficulties, and general weakness. The maximum tolerated dose of cisplatin is around 100 mg per day for up to only five consecutive days. While the drug displays high activity, these unfortunate side- effects led to a search for a better analogue (Figure 9.3). Carboplatin [cis-diammine (1.1- cyclobutanedicarboxylato) platinum (II)] was introduced as a second-generation drug in 1990. It shows about a fourfold reduction in side effects to the kidneys and nervous system. Because the drugs must circulate in the bloodstream before finding their target, slower degradation in vivo is believed to be one factor of importance. Both cisplatin and carboplatin suffer from the emergence of acquired resistance with time. Subsequently, oxaliplatin [(1,2- cyclohexane diamine) (oxalato)platinum (II)] was introduced as a third-generation drug. It is active in all phases of the cell cycle, and binds covalently to DNA guanine and adenine bases dominantly via intrastrand crosslinking. It is thought that DNA mismatch repair enzymes are unable to recognize oxaliplatin-DNA adducts in contrast with some other platinum-DNA adducts, as a result of the bulkier nature of the complex. The complex is inactivated by thiol protein binding, alteration in cellular transport and increased DNA repair enzyme activity. Development of a new-generation drug to address inactivation has led to satr a plat in the
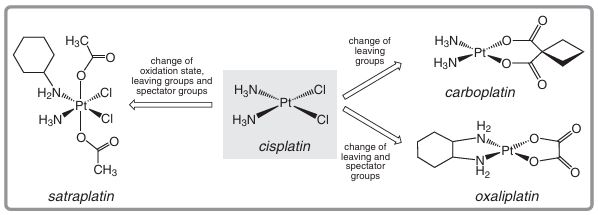
Figure 9.3 Evolution of the first-generation platinum drug cisplatin into later generation drugs.
first orally-administrable platinum drug - in this case a platinum (IV) complex. A mode of action that involves reduction to Pt(II) in vivo has been proposed. This drug shows no neurotoxicity or nephrotoxicity, with bone marrow supression the dose-limiting adverse effect; it is under consideration for approval as a drug in prostrate cancer treatment.
Platinum complexes are currently the best-selling anticancer drugs in the world, with billion-dollar annual sales. Nevertheless the search for better platinum drugs goes on including development of some polynuclear complexes. Chemotherapy with platinum drugs can be effective, but may have severe side effects because the drugs are toxic and cannot discriminate sufficiently between normal and cancer cells. Development of new drugs centred on the introduction of components that deliver the drugs specifically to cancer cells will assist in specificity and reduction of side effects.
 الاكثر قراءة في الكيمياء التناسقية
الاكثر قراءة في الكيمياء التناسقية
 اخر الاخبار
اخر الاخبار
اخبار العتبة العباسية المقدسة

الآخبار الصحية















 قسم الشؤون الفكرية يصدر كتاباً يوثق تاريخ السدانة في العتبة العباسية المقدسة
قسم الشؤون الفكرية يصدر كتاباً يوثق تاريخ السدانة في العتبة العباسية المقدسة "المهمة".. إصدار قصصي يوثّق القصص الفائزة في مسابقة فتوى الدفاع المقدسة للقصة القصيرة
"المهمة".. إصدار قصصي يوثّق القصص الفائزة في مسابقة فتوى الدفاع المقدسة للقصة القصيرة (نوافذ).. إصدار أدبي يوثق القصص الفائزة في مسابقة الإمام العسكري (عليه السلام)
(نوافذ).. إصدار أدبي يوثق القصص الفائزة في مسابقة الإمام العسكري (عليه السلام)